How Do You Know When to Use Stereochem
Stereochemistry of the Diels-Alder Reaction
Here's what we've learned near the Diels Alder reaction so far: [previous post in this series]
- iii pi bonds are ever cleaved
- two sigma bonds and a pi bail are always formed, resulting in a new vi-membered ring
- electron withdrawing groups on the dienophile increase the reaction rate
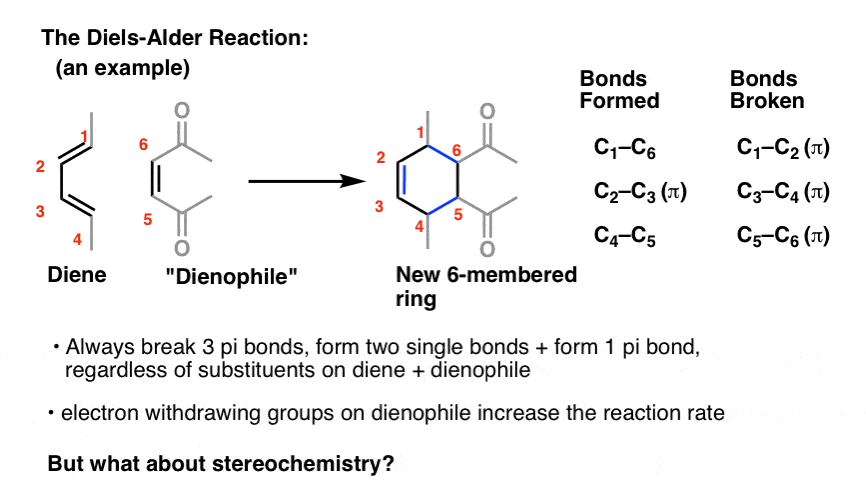
One cistron we oasis't addressed yet? Stereochemistry.
Let'due south get started!
Tabular array of Contents
- Stereochemistry In The Diels-Alder: A Tale of Two Dienophiles (cis– andtrans-)
- The Relationship Of The Substitutents Nigh The Double Bail In The Dienophile Is Preserved In The Diels-Alder Product
- Stereochemistry of The Diene Substituents In The Diels-Alder Product: "Outside" And "Within" Substituents
- Applying The "Exterior / Within" Rule For Dienes
- What Happens When Both the Diene and Dienophile Are Substituted?
- When Substituted Dienes React With Substituted Dienophiles, Diastereomers May Also Be Formed (Note: We Call These "Exo" and "Endo")
- Endo And Exo Diels-Alder Products Are Diastereomers
- Summary: Stereochemistry of the Diels-Alder Reaction
- Notes
1. Stereochemistry In The Diels-Alder: A Tale of Two Dienophiles (cis– andtrans-)
A pi bond is broken on the dienophile during the course of the Diels-Alder reaction, and the hybridization goes from sp2 to sp3. So what happens to the stereochemistry of the groups attached the pi bond?
Take the 2 dienophiles maleic acid and fumaric acrid for example. These two molecules are diastereomers, differing only in the orientation of the ii carboxylic acid groups about the double bail.
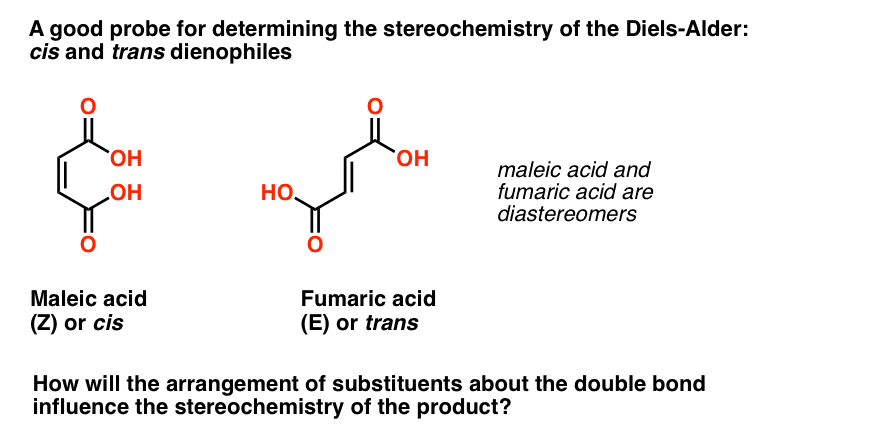
This pair of diastereomers makes an splendid probe for determining how the stereochemistry of the dienophile pi bond is affected in the Diels-Alder reaction.
Will thecis carboxylic acids of maleic acid remaincis in the production? Will thetrans carboxylic acids of fumaric acid remaintransin the product? Or does something else happen?
Here's what experiments show us:
2. The Relationship Of The Substitutents About The Double Bond In The Dienophile Is Preserved In The Diels-Alder Product
Let's call this Diels-Alder Stereochemistry Rule #1:
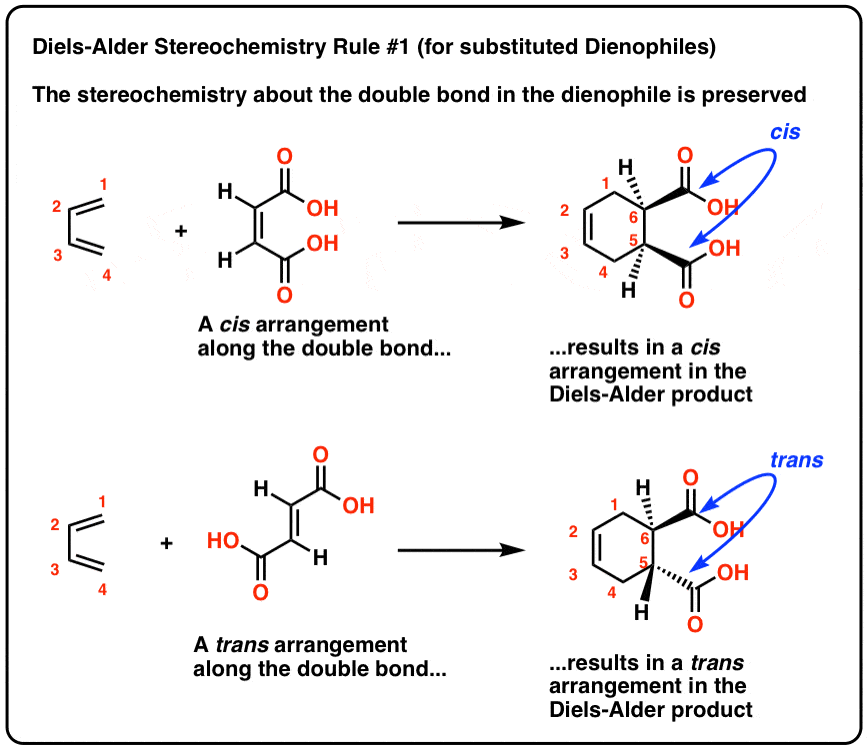
cis– dienophiles requite u.s.cis-products, andtrans– dienophiles give ustrans-products.
Aught sneaky, in other words.
What about the diene?
three. Stereochemistry of The Diene Substituents In The Diels-Alder Product: "Exterior" And "Inside" Substituents
Substituents on C-2 and C-iii of the diene aren't an issue: they start the reaction on a (flat) sp2 hybridized carbon and end the reaction on a (flat) sptwo hybridized carbon. No chiral centers are created here, and so in that location's no stereochemistry issues to concern ourselves with.
Simply what most the substituents on C-1 and C-4? They are on an sptwo hybridized carbon in the starting fabric and end upwards on an sp3 hybridized carbon in the product.
Here's what we observe from experiment:
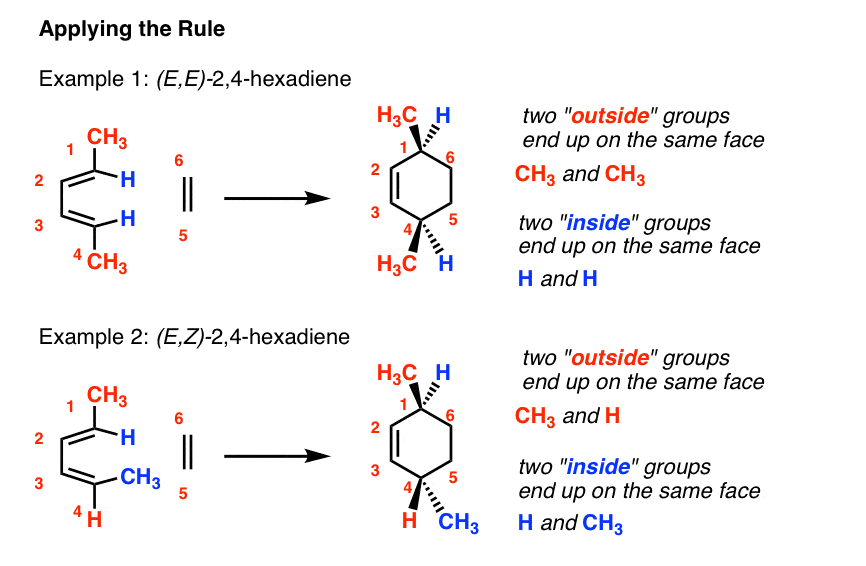
It turns out that the two "exterior groups" on the diene (labelled "A", beneath) when fatigued in thedue south-cis conformation end up on 1 face of the new six-membered band, and the 2 "inside" groups (labelled "B") both cease up on the other confront of the ring. Let's call this Diels-Alder Stereochemistry Rule #2.
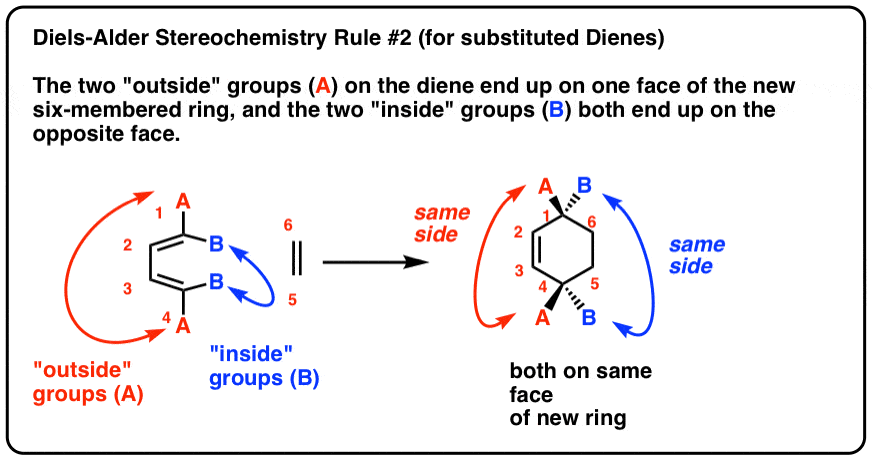
For case, let'south examine ii isomers of two,4 hexadiene: (E, E) [example 1] and (Eastward, Z) [example 2].
four. Applying The "Exterior / Inside" Rule For Dienes
Cartoon each diene in thesouthward–cis conformation, which is necessary for the Diels-Alder to proceed, we run into that the ii "exterior" groups end upwards on the same face up of the six-membered ring, and the two "inside" groups as well end upwards on the same face of the 6-membered ring.
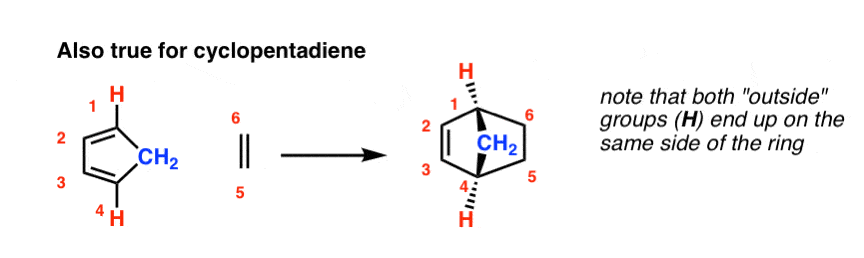
Actually, if we expect back to ane of the primeval examples of the Diels-Alder that we've seen, this is also truthful for cyclopentadiene:
And then far, I hope that this seems straightforward enough.
And then allow's combine these two effects, and meet what happens!
5. What Happens When Both the Diene and Dienophile Are Substituted?
If we accept substitution onboththe diene and dienophile, what happens? What do we do then?
The same matter! Rule #1 and Rule #2 nevertheless hold. They concord for every single Diels-Alder reaction, actually.
For case, here'southward the example of the reaction of fumaric acrid with (E, E)2,four hexadiene:
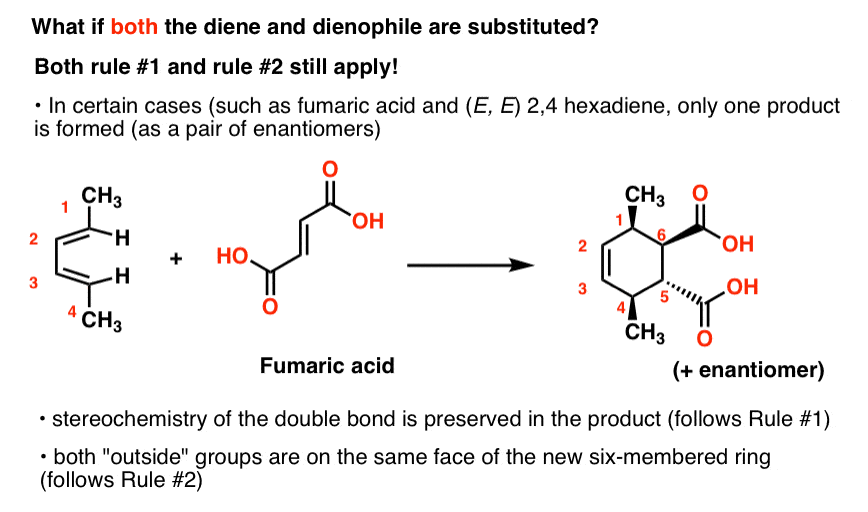
This results in a single production formed as a racemic mixture of enantiomers.
Simple?
Non always.
half dozen. When Substituted Dienes React With Substituted Dienophiles, Diastereomers May Also Be Formed (Note: We Call These "Exo" and "Endo")
Fumaric acid has the belongings of beingness symmetrical with respect to rotation (yous might sometimes hear this described equally C2 symmetry), which has the consequence that but one production (equally a pair ofenantiomers) is formed in the reaction with 2,four-hexadiene.
However, fumaric acid'due south cousin, maleic acid, lacks this belongings.
When we combine a substituted diene such equally (E, E) 2,4-hexadiene with maleic acrid, and follow both Dominion #1 and Dominion #2, in that location are reallytwo possible products!
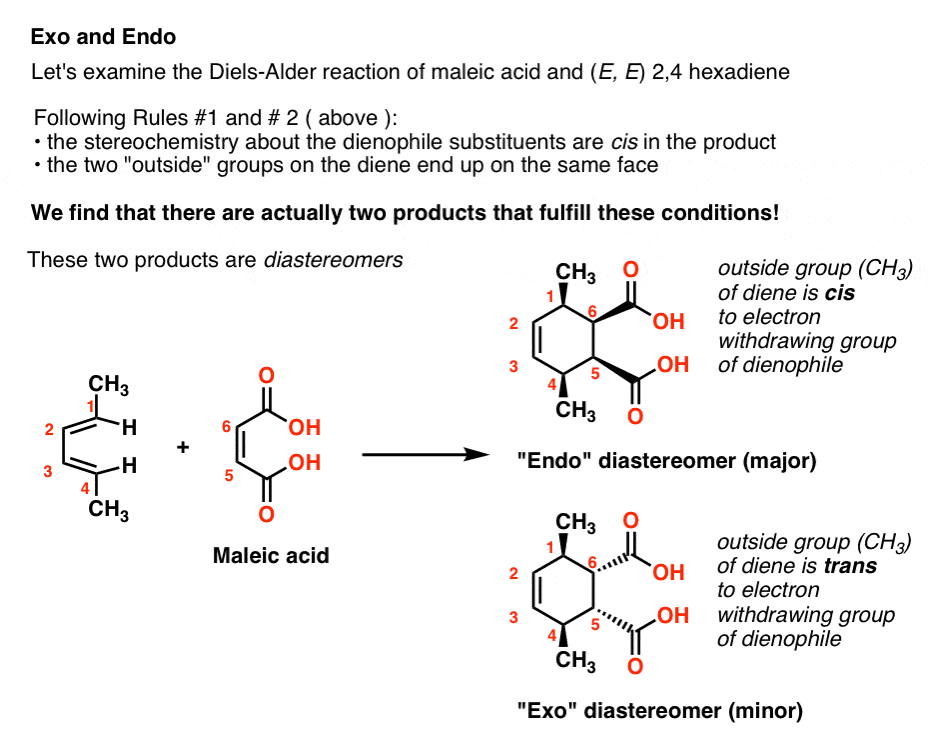
In the first product, the "outside" CHthree groups are on the aforementioned side of the new six-membered band equally the carboxylic acids in maleic acid. We call this the "endo" product.
In the 2d product, the "outside" CH3 groups are on the contrary side of the new six-membered ring as the carboxylic acids in maleic acid. We call this the "exo" production.
7. Endo And Exo Diels-Alder Products Are Diastereomers
The "endo" and "exo" products in this case arediastereomers.They are stereoisomers of each other, but are not enantiomers. (In fact, neither the "endo" or "exo" products in the instance above possess an enantiomer. Can yous run across why?).
In practice, for reasons that will not exist immediately obvioius, the "endo" product tends to exist favored over the "exo" product.
This subject area of exo and endo turns out to be such an important topic that it deserves its own article. So we'll explore exactly how to tell the departure between "exo" and "endo" products, as well every bit how they course, in the side by side article.
8. Summary: Stereochemistry of the Diels-Alder Reaction
- The stereochemistry of the dienophile is preserved in the Diels-Alder production
- The "outside" groups on the diene end upwardly on the same confront of the new vi-membered ring, as do the "inside" groups.
- When both the diene and dienophile are substituted, diastereomers may form, which we call "exo" and "endo".
Thanks to Tom Struble for help with this post.
Notes
(One last note. The Diels-Alder reaction of (E,E) 2,4 hexadiene with fumaric acid produced a pair of enantiomers, but neither of the products of the Diels-Alder of (E,E)-2,4-hexadiene with maleic acid has an enantiomer. Can you meet why?)
How Do You Know When to Use Stereochem
Source: https://www.masterorganicchemistry.com/2017/11/13/stereochemistry-of-the-diels-alder-reaction/
0 Response to "How Do You Know When to Use Stereochem"
Post a Comment